 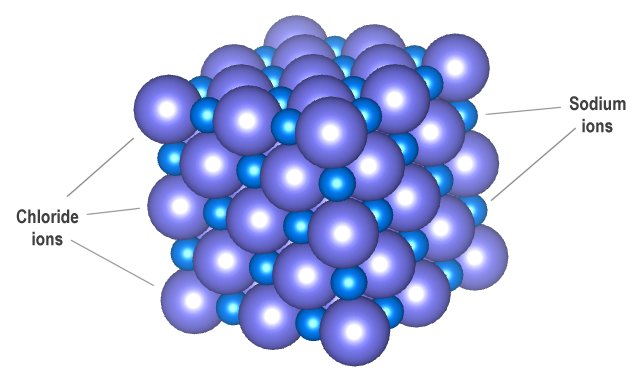
Eventually, the whole surface consists of smooth, stable faces, where new atoms cannot as easily attach themselves.Ĭrystals are commonly recognized, macroscopically, by their shape, consisting of flat faces with sharp angles. Therefore, these parts of the crystal grow out very quickly (yellow arrows). These are grouped into 7 crystal systems, such as cubic crystal system (where the crystals may form cubes or rectangular boxes, such as halite shown at right) or hexagonal crystal system (where the crystals may form hexagons, such as ordinary water ice).Īs a halite crystal is growing, new atoms can very easily attach to the parts of the surface with rough atomic-scale structure and many dangling bonds. There are 219 possible crystal symmetries (230 is commonly cited, but this treats chiral equivalents as separate entities), called crystallographic space groups. The symmetry of a crystal is constrained by the requirement that the unit cells stack perfectly with no gaps. The unit cells are stacked in three-dimensional space to form the crystal. There are distinct differences between crystalline solids and amorphous solids: most notably, the process of forming a glass does not release the latent heat of fusion, but forming a crystal does.Ī crystal structure (an arrangement of atoms in a crystal) is characterized by its unit cell, a small imaginary box containing one or more atoms in a specific spatial arrangement. These have no periodic order, even microscopically. Solids that are neither crystalline nor polycrystalline, such as glass, are called amorphous solids, also called glassy, vitreous, or noncrystalline. Most macroscopic inorganic solids are polycrystalline, including almost all metals, ceramics, ice, rocks, etc. In the final block of ice, each of the small crystals (called " crystallites" or "grains") is a true crystal with a periodic arrangement of atoms, but the whole polycrystal does not have a periodic arrangement of atoms, because the periodic pattern is broken at the grain boundaries. For example, when liquid water starts freezing, the phase change begins with small ice crystals that grow until they fuse, forming a polycrystalline structure. ( Quasicrystals are an exception, see below). A crystal is a solid where the atoms form a periodic arrangement. 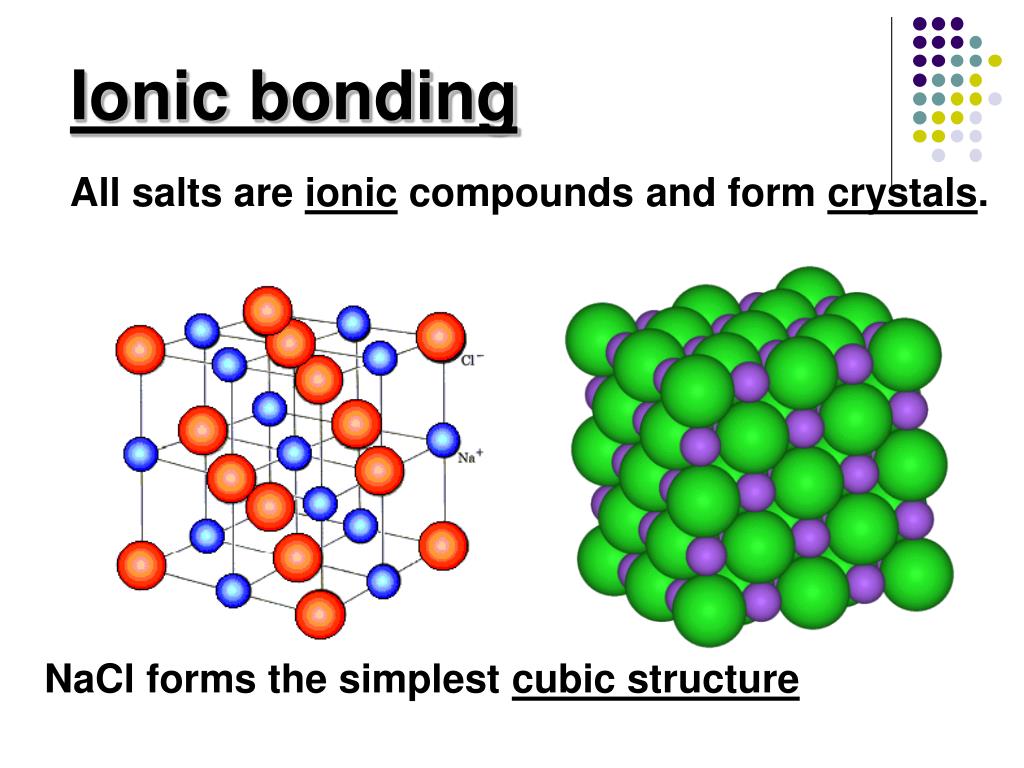
The scientific definition of a "crystal" is based on the microscopic arrangement of atoms inside it, called the crystal structure. amorphous solids.Ĭrystals, or crystalline solids, are often used in pseudoscientific practices such as crystal therapy, and, along with gemstones, are sometimes associated with spellwork in Wiccan beliefs and related religious movements. Examples of amorphous solids include glass, wax, and many plastics.ĭespite the name, lead crystal, crystal glass, and related products are not crystals, but rather types of glass, i.e. 
A third category of solids is amorphous solids, where the atoms have no periodic structure whatsoever. Polycrystals include most metals, rocks, ceramics, and ice. many microscopic crystals fused together into a single solid. Most inorganic solids are not crystals but polycrystals, i.e. Įxamples of large crystals include snowflakes, diamonds, and table salt. The word crystal derives from the Ancient Greek word κρύσταλλος ( krustallos), meaning both " ice" and " rock crystal", from κρύος ( kruos), "icy cold, frost". The process of crystal formation via mechanisms of crystal growth is called crystallization or solidification. The scientific study of crystals and crystal formation is known as crystallography. In addition, macroscopic single crystals are usually identifiable by their geometrical shape, consisting of flat faces with specific, characteristic orientations. Microscopically, a single crystal has atoms in a near-perfect periodic arrangement a polycrystal is composed of many microscopic crystals (called " crystallites" or "grains") and an amorphous solid (such as glass) has no periodic arrangement even microscopically.Ī crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
